Bioplasmonics Group
Publications
The ability to spatially and temporally map nanoscale environments in situ over extended time scales would be transformative for biology, biomedicine, and bioengineering. All nanometer objects, from nanoparticles down to single proteins, scatter light. Interferometric scattering stands as a powerful tool, offering ultrasensitivity and resolution vital for visualizing nanoscale entities. Interferometric scattering from an individual nanoparticle down to an individual protein has been detected; however, resolving adjacent nanometer objects with interferometric scattering has not yet been demonstrated. In this work, we present interferometric phase intensity nanoscopy (iPINE) to resolve adjacent nanometer objects with interferometric scattering. We demonstrate that multiphase and sensitivity of iPINE reveal ellipse Airy patterns correlated with nanostructural features. We show that eliminating background fluctuation by employing circularly polarized illumination in iPINE is essential to separate proximal nanometer objects below the diffraction limit. We envision iPINE for resolving a variety of adjacent nanometer objects from nanoparticles down to proximal proteins in situ over extended time periods for a wide range of applications in biology, biomedicine, and bioengineering. We expect iPINE to be especially important in applications of biological dynamics that require extended observation times.
Fundamental to all living organisms and living soft matter are emergent processes in which the reorganization of individual constituents at the nanoscale drives group-level movements and shape changes at the macroscale over time. However, light-induced degradation of fluorophores, photobleaching, is a significant problem in extended bioimaging in life science. Here, we report opening a long-time investigation window by nonbleaching phase intensity nanoscope: PINE. We accomplish phase-intensity separation such that nanoprobe distributions are distinguished by an integrated phase-intensity multilayer thin film (polyvinyl alcohol/liquid crystal). We overcame a physical limit to resolve sub-10 nm cellular architectures, and achieve the first dynamic imaging of nanoscopic reorganization over 250 h using PINE. We discover nanoscopic rearrangements synchronized with the emergence of group-level movements and shape changes at the macroscale according to a set of interaction rules with importance in cellular and soft matter reorganization, self-organization, and pattern formation.
Precise nanostructure geometry that enables the optical biomolecular delivery of nanosensors to the living intracellular environment is highly desirable for precision biological and clinical therapies. However, the optical delivery through membrane barriers utilizing nanosensors remains difficult due to a lack of design guidelines to avoid inherent conflict between optical force and photothermal heat generation in metallic nanosensors during the process. Here, we present a numerical study reporting significantly enhanced optical penetration of nanosensors by engineering nanostructure geometry with minimized photothermal heating generation for penetrating across membrane barriers. We show that by varying the nanosensor geometry, penetration depths can be maximized while heat generated during the penetration process can be minimized. We demonstrate the effect of lateral stress induced by an angularly rotating nanosensor on a membrane barrier by theoretical analysis. Furthermore, we show that by varying the nanosensor geometry, maximized local stress fields at the nanoparticle–membrane interface enhanced the optical penetration process by four-fold. Owing to the high efficiency and stability, we anticipate that precise optical penetration of nanosensors to specific intracellular locations will be beneficial for biological and therapeutic applications.
Plasmonic nanostructures, functioning as nano-antenna reactors to highly focus light and efficiently convert it into thermal/chemical energy, have a significant potential for sustainable solar water disinfection (SODIS). A high-density plasmonic nanogap-based reactor array opens a way of maximizing the photothermal effect, but achieving uniformity and large density in such a technology while scaling up remains challenging. In this study, we provide an integrative plasmonic dimer array (hPDA) that is uniform and has high density ensuring significantly enhanced SODIS performance. The hPDA is constructed by a combined fabrication of the self-assembled monolayer and block copolymer lithography approaches. This combination leads to a two-dimensional hexagonal array of the Au dimer structures consisting of a 1.3 nm nanogap. The uniformity and high density of nanogaps of the hPDA result in the physically and optically stabilized dimer array, which allows strong light focusing and a rapid and highly efficient harvesting of photothermal energy in the visible region. Finally, the integrated hPDA with an optofluidic reactor enables 5-fold enhanced Escherichia coli (E. coli) disinfection. We anticipate that the hPDA will be useful in the scalable sustainable energy and environmental process that converts solar energy.
Dynamic detection in challenging lighting environments is essential for advancing intelligent robots and autonomous vehicles. Traditional vision systems are prone to severe lighting conditions in which rapid increases or decreases in contrast or saturation obscures objects, resulting in a loss of visibility. By incorporating intelligent optimization of polarization into vision systems using the iNC (integrated nanoscopic correction), we introduce an intelligent real-time fusion algorithm to address challenging and changing lighting conditions. Through real-time iterative feedback, we rapidly select polarizations, which is difficult to achieve with traditional methods. Fusion images were also dynamically reconstructed using pixel-based weights calculated in the intelligent polarization selection process. We showed that fused images by intelligent polarization selection reduced the mean-square error by two orders of magnitude to uncover subtle features of occluded objects. Our intelligent real-time fusion algorithm also achieved two orders of magnitude increase in time performance without compromising image quality. We expect intelligent fusion imaging photonics to play increasingly vital roles in the fields of next generation intelligent robots and autonomous vehicles.
The crowded intracellular environment of biomolecules, including organelles, solutes, proteins, and membranes, presents distinct biomolecular dynamics crucial for the functions of biomolecules within living cells. However, background suppression is critical to uncover nanoscale dynamics in living cells. Herein, a new method for enabling rapid, nanoscale background elimination of cellular metallic nanoprobes is presented. By employing integrated nanoscopic correction (iNC) designed to eliminate depolarization effects, which compromise background elimination, a real-time algorithm to subtract and increment orthogonal pairs of polarizations for real-time nanoscale background elimination is introduced. The ability to analyze orthogonal pairs at high speed over the entire polarization range is currently difficult to achieve using conventional methods. By processing orthogonal pairs in real time, this method minimizes movement artifacts during the background elimination process. Nanometer spatial stability which enables two orders of magnitude increase in signal-to-noise ratio of cellular metallic nanoprobes is shown. Nanoscale background elimination aiding the ability to accurately track biomolecules and their dynamics in living cells is anticipated.
CRISPR-Cas systems (clustered regularly interspaced short palindromic repeats) have shown great potential as efficient gene editing tools in disease therapeutics. Although numerous CRISPR-Cas systems have been developed, detailed mechanisms of target recognition and DNA cleavage are still unclear. In this work, we dynamically observe the entire process of conjugation, target recognition and DNA cleavage by single particle spectroscopy of CRISPR-Cas systems on single particle surfaces (gold) with the unique advantage of extended time periods. We show the CRISPR-Cas system, comprised of Cas endonuclease and single guide RNA, is stable and functional on single particle surfaces. Owing to the photostability of single particle surfaces, we directly observe in real time the entire dynamic process of conjugation, target recognition and DNA cleavage without photobleaching. We find heterogeneity in target recognition and DNA cleavage processes in which individual spectra vary significantly from one another as well as from the ensemble. We believe an in depth understanding of heterogeneities in CRISPR-Cas systems can overcome potential barriers in precision medicine and personalized disease therapeutics.
Multifunctional nanoprobes have attracted significant attention in a wide range of disciplines such as nanomedicine, precision medicine, and cancer diagnosis and treatment. However, integrating multifunctional ability in a nanoscale structure to precisely target, image, and deliver with cellular spatial/temporal resolution is still challenging in cellulo applications. This is because the development of such high-precision resolution needs to be carried out without labeling, photobleaching, and structurally segregating live cells. In this study, we present an integrated nanostructure of a mesoporous-silica nanosphere with an optical nanocrescent antenna (MONA) for multifunctional cellular targeting, drug delivery, and molecular imaging with spatiotemporal resolution. MONA comprises a systematically constructed Au nanocrescent (AuNC) antenna as a nanosensor and optical switch on a mesoporous-silica nanosphere as a cargo to molecular delivery. MONA made of antiepithelial cell adhesion molecules (anti-EpCAM)-conjugated AuNC facilitates the specific targeting of breast cancer cells, resulting in a highly focused photothermal gradient that functions as a molecular emitter. This light-driven molecular, doxorubicin (DOX) delivery function allows rapid apoptosis of breast cancer cells. Since MONA permits the tracking of quantum biological electron-transfer processes, in addition to its role as an on-demand optical switch, it enables the monitoring of the dynamic behavior of cellular cytochrome c pivoting cell apoptosis in response to the DOX delivery. Owing to the integrated functions of molecular actuation and direct sensing at the precisely targeted spot afforded by MONA, we anticipate that this multifunctional optical nanoantenna structure will have an impact in the fields of nanomedicine, cancer theranostics, and basic life sciences.
Targeted delivery of drugs to tumor cells, which circumvent resistance mechanisms and induce cell killing, is a lingering challenge that requires innovative solutions. Here, we provide two bioengineered strategies in which nanotechnology is blended with cancer medicine to preferentially target distinct mechanisms of drug resistance. In the first ‘case study’, we demonstrate the use of lipid–drug conjugates that target molecular signaling pathways, which result from taxane-induced drug tolerance via cell surface lipid raft accumulations. Through a small molecule drug screen, we identify a kinase inhibitor that optimally destroys drug tolerant cancer cells and conjugate it to a rationally-chosen lipid scaffold, which enhances anticancer efficacy in vitro and in vivo. In the second ‘case study’, we address resistance mechanisms that can occur through exocytosis of nanomedicines. Using adenocarcinoma HeLa and MCF-7 cells, we describe the use of gold nanorod and nanoporous vehicles integrated with an optical antenna for on-demand, photoactivation at ∼650 nm enabling release of payloads into cells including cytotoxic anthracyclines. Together, these provide two approaches, which exploit engineering strategies capable of circumventing distinct resistance barriers and induce killing by multimodal, including nanophotonic mechanisms.
Optical manipulation and imaging of nano-objects with nanometer precision is highly desirable for nanomaterial and biological studies due to inherent noninvasiveness. However, time constraints and current segregated experimental systems for nanoimaging and nanomanipulation limits real-time super-resolution imaging with spatially enhanced manipulation. Here, an integrated nanoscopic correction (iNC) method to enable multimodal nanomanipulation-nanoimaging is reported. The iNC consists of a multimodal voltage-tunable power modulator, polarization rotator, and polarizer. Using the iNC, plasmonic nano-objects which are below the diffraction limit and which can be distinguished by direct observation without post processing are demonstrated. Furthermore, such direct observations with enhanced nanometer spatial stability and millisecond high speed are shown. Precise trapping and rapid rotation of gold nanorods with the iNC are demonstrated successfully. With non-invasive post-processing free nanoimaging and nanomanipulation, it is anticipated that the iNC will make contributions in the nanomaterial and biological sciences requiring precision optics.
The development of sustainable methods for energy-intensive water treatment processes continues to be a challenging issue. Plasmonic-semiconductor nanoparticles, which absorb large amounts of sunlight in the visible range for conversion into chemical energy efficiently, can form the basis of a sustainable water treatment method. However, the potential uses of plasmonic semiconductor particles for water treatment have not been fully explored yet because of the limitations associated with the imbalance between light capture, charge transfer, and the required recycling steps for the particles themselves. Herein, a significantly improved visible-light-induced water treatment method that uses a plasmo-semiconductor nanogap bridge array (PNA) is reported. As an arrangement of antenna-reactors, the PNA enables the balancing of the largely enhanced electromagnetic field in the plasmonic nanogap coupling region and optimal separation of charge carriers in the semiconductor. The simultaneous effects of visible-light absorption and charge transfer lead to the generation of a highly enhanced visible-light-induced OH radical (•OH). Consequently, visible-light-induced 5-log N/N0 water disinfection and 100% chemical decomposition for sustainable water treatment were demonstrated. Owing to the large light absorption, charge carrier utilization, and array-oriented scalability, the PNA will be valuable in various sustainable energy and environmental applications.
Visualization of dynamic interlinking networks which respond and adapt to the constantly changing environment would be highly beneficial in developing new composite materials and active/responsive materials. Here, optically and structurally stabilized plasmo-bio interlinking networks (PBINs) free from photobleaching for high resolution, long term visualization are reported. Necessary for structural and optical stability, a new stability algorithm to comprehensively quantify stability and detect minute instability undetectable by traditional methods is introduced. Biocompatible plasmonic gold nanorods (Bio-AuNRs) are synthesized for high resolution, long term imaging by utilizing bromide-free alternatives to achieve CTA+ free. Systematic physical, chemical, and biological characterizations reveal the structural and optical stability of Bio-AuNRs required for constructing PBIN. Lastly, with actin as a model of interlinking networks of the cytoskeleton, optically and structurally stable PBIN (100% CTA+ free, 97% crosslinking rate) in applications as active/responsive materials, are demonstrated.
Conventional biological imaging typically relies on either spatial or temporal modulation which are limited by fluorophores undergoing photobleaching. As a result, imaging of biological processes for extended periods of time remains a challenge. To overcome this limitation, we introduce polarization modulation combined with nonbleaching plasmonic nanoparticles to boost signal-to-noise ratio and to eliminate strong background scattering. We show plasmonic nanoparticles can be extracted from the noisy background and cellular environment, where the visualization of nanoparticles has been previously hindered by the strongly scattering biological components. We demonstrate that such a reduction of background noise level greatly enhances the overall signal-to-noise ratio of nanoparticles within biological structures.
Plasmonic nanoparticles are expected to impact various fields, such as chemical analysis, superresolution imaging, and therapeutics. However, since plasmonic nanoparticles are known to be prone to aggregation, widespread use of plasmonic nanoparticles is still limited. While many methods have been proposed to enhance colloidal stability, a universal method to comprehensively detect stability is still lacking. We present a new comprehensive stability parameter (CSP) as a robust and universal method to quantify stability. Unlike other methods, CSP utilizes the entire UV-Vis spectrum to evaluate the aggregation and to avoid potential biasing issues by only looking at a single parameter. We quantitatively analyzed the colloidal stability of plasmonic particles with different surface coatings using CSP. This work establishes a standardized quantification method which can be used in the future nanoparticle applications.
Plasmonic nanoparticles are excellent nonbleaching probes for bio-imaging. Due to their anisotropic properties, polarization analysis of individual nanoparticles allows for revealing orientational information, plasmon mode assignment, and the local microenvironment. Previous implementations utilize mechanical rotation of conventional polarizers to align the polarization angles with specific axes of nanoparticles. However, the manufacturing defects of the polarizer (e.g., non-parallelism) limit the measurement stability (e.g., beam wobbling) in polarimetric imaging, while the mechanical rotation limits the measurement speed, and thus hinders accurate, real-time acquisition of individual nanoparticles. Here, we demonstrate a high-speed nano-polarimetric system for stable plasmonic bio-imaging by integrating our voltage-tunable polarizer (VTP) into a microscope. The angular rotation of the polarization (0~π) can be realized by applying voltage on the VTP. We show that our voltage-tunable system offers high extinction ratio (~up to 250), and uniform transmission (~55%) over a large input power range (less than 5% deviation for input power from 50 µW to ~20 mW). Meanwhile, the transmission polarization can be rapidly tuned with a response time up to 50 ms. Compared to conventional polarizers, our system is able to provide reproducible and high-speed polarimetric images of individual nanoparticles with sub-pixel spatial precision. Such a polarimetric nanoimaging system could be a useful tool for real-time single nanoparticle bio-imaging with both high stability and time resolution.
Precise polarimetric imaging of polarization-sensitive nanoparticles is essential for resolving their accurate spatial positions beyond the diffraction limit. However, conventional technologies currently suffer from beam deviation errors which cannot be corrected beyond the diffraction limit. To overcome this issue, we experimentally demonstrate a spatially stable nano-imaging system for polarization-sensitive nanoparticles. In this study, we show that by integrating a voltage-tunable imaging variable polarizer with optical microscopy, we are able to suppress beam deviation errors. We expect that this nano-imaging system should allow for acquisition of accurate positional and polarization information from individual nanoparticles in applications where real-time, high precision spatial information is required.
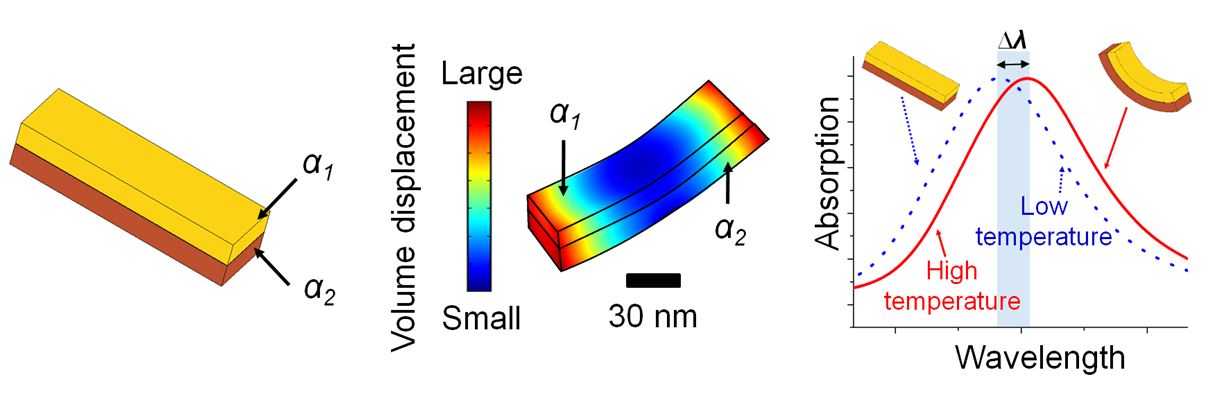
We propose and theoretically demonstrate a mechano-optical nano-antenna over a broad temperature range. We show that there is a tunable, temperature-dependent plasmonic resonance associated with the nano-antenna geometry. We also theoretically demonstrate a matching condition for mechanical properties that is essential for maximizing thermal expansion differences across a broad temperature range. We expect that mechano-optical ano-antennas should allow for spatiotemporal temperature mapping in applications where precise measurement of local temperature is needed in real time.
** Highlighted in J. Lab Autom., 2016, 21(2), 227-233**
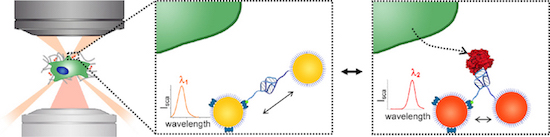
Plasmon rulers, consisting of pairs of gold nanoparticles, allow single-molecule analysis without photobleaching or blinking; however, current plasmon rulers are irreversible, restricting detection to only single events. Here, we present a reversible plasmon ruler, comprised of coupled gold nanoparticles linked by a single aptamer, capable of binding individual secreted molecules with high specificity. We show that the binding of target secreted molecules to the reversible plasmon ruler is characterized by single-molecule sensitivity, high specificity, and reversibility. Such reversible plasmon rulers should enable dynamic and adaptive live-cell measurement of secreted single molecules in their local microenvironment.
Spatiotemporal activity patterns of proteases such as matrix metalloproteinases and cysteine proteases in organs have the potential to provide insight into how organized structural patterns arise during tissue morphogenesis and may suggest therapeutic strategies to repair diseased tissues. Toward imaging spatiotemporal activity patterns, recently increased emphasis has been placed on imaging activity patterns in three-dimensional culture models that resemble tissues in vivo. Here, we briefly review key methods, based on fluorogenic modifications either to the extracellular matrix or to the protease-of-interest, that have allowed for qualitative imaging of activity patterns in three-dimensional culture models. We highlight emerging plasmonic methods that address significant improvements in spatial and temporal resolution and have the potential to enable quantitative measurement of spatiotemporal activity patterns with single-molecule sensitivity.
** Selected as cover article**
** Highlighted in ACS Nano, 2012, 6(9), 7548-7552**
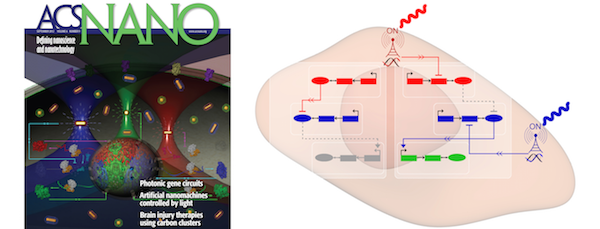
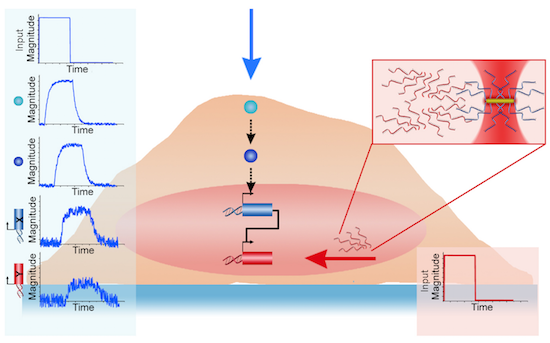
The precise perturbation of gene circuits and the direct observation of signaling pathways in living cells are essential for both fundamental biology and translational medicine. Current optogenetic technology offers a new paradigm of optical control for cells; however, this technology relies on permanent genomic modifications with light-responsive genes, thus limiting dynamic reconfiguration of gene circuits. Here, we report precise control of perturbation and reconfiguration of gene circuits in living cells by optically addressable siRNA-Au nanoantennas. The siRNA-Au nanoantennas fulfill dual functions as selectively addressable optical receivers and biomolecular emitters of small interfering RNA (siRNA). Using siRNA-Au nanoantennas as optical inputs to existing circuit connections, photonic gene circuits are constructed in living cells. We show that photonic gene circuits are modular, enabling subcircuits to be combined on-demand. Photonic gene circuits open new avenues for engineering functional gene circuits useful for fundamental bioscience, bioengineering, and medical applications.
This review focuses on the recent developments in nanoplasmonic gene regulations. Types of nanoplasmonic carriers and DNA/RNA cargo are described. Strategies to liberate cargo from their carriers using NIR and enable on-demand silencing of endogenous intracellular genes are reviewed. In addition to inhibitory effects, exogenous foreign genes are also introduced and expressed on-demand using nanoplasmonic optical switches. The magnitude and timing of genetic activities can therefore be systematically controlled on-demand remotely. Equipped with new nanoplasmonic optics to directly probe the intracellular space, quantitative approaches should capture many dynamic activities within living systems that were otherwise previously impossible to control using conventional methods.
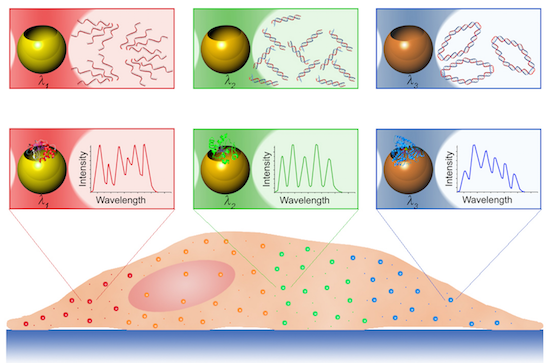
Free electrons in a noble metal nanoparticle can be resonantly excited, leading to their collective oscillation termed as a surface plasmon. These surface plasmons enable nanoparticles to absorb light, generate heat, transfer energy, and re-radiate incident photons. Creative designs of nanoplasmonic optical antennae (i.e. plasmon resonant nanoparticles) have become a new foundation of quantitative biology and nanomedicine. This review focuses on the recent developments in dual-functional nanoplasmonic optical antennae for label-free biosensors and nanoplasmonic gene switches. Nanoplasmonic optical antennae, functioning as biosensors to significantly enhance biochemical-specific spectral information via plasmon resonance energy transfer (PRET) and surface-enhanced Raman spectroscopy (SERS), are discussed. Nanoplasmonic optical antennae, functioning as nanoplasmonic gene switches to enable spatiotemporal regulation of genetic activity, are also reviewed. Nanoplasmonic molecular rulers and integrated photoacoustic–photothermal contrast agents are also described.
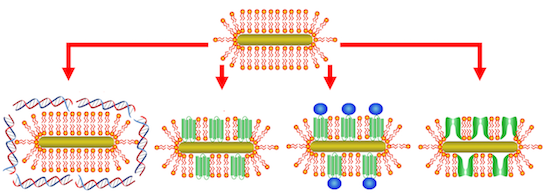
Biologically functional cationic phospholipid−gold nanoplasmonic carriers have been designed to simultaneously exhibit carrier capabilities, demonstrate improved colloidal stability, and show no cytotoxicity under physiological conditions. Cargo, such as RNA, DNA, proteins, or drugs, can be adsorbed onto or incorporated into the cationic phospholipid bilayer membrane. These carriers are able to retain their unique nanoscale optical properties under physiological conditions, making them particularly useful in a wide range of imaging, therapeutic, and gene delivery applications that utilize selective nanoplasmonic properties.
** Highlighted in Nature Photonics, 2009, 3, 126-127**
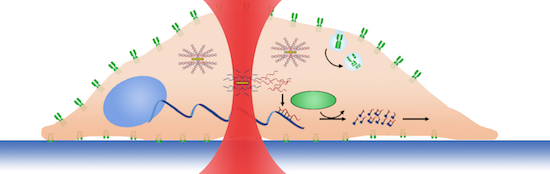
Near infrared-absorbing gold nanoplasmonic particles (GNPs) are used as optical switches of gene interference and are remotely controlled using light. We have tuned optical switches to a wavelength where cellular photodamage is minimized. Optical switches are functionalized with double-stranded oligonucleotides. At desired times and at specific intracellular locations, remote optical excitation is used to liberate gene-interfering oligonucleotides. We demonstrate a novel gene-interfering technique offering spatial and temporal control, which is otherwise impossible using conventional gene-interfering techniques.
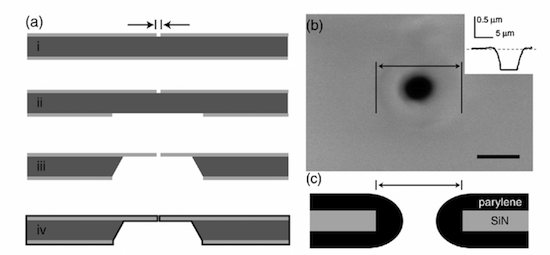
We report here a non-invasive, reversible method for interrogating single cells in a microfluidic flow-through system. Impedance spectroscopy of cells held at a micron-sized pore under negative pressure is demonstrated and used to determine the presence and viability of the captured cell. The cell capture pore is optimized for electrical response and mechanical interfacing to a cell using a deposited layer of parylene. Changes in the mechanical interface between the cell and the chip due to chemical exposure or environmental changes can also be assessed. Here, we monitored the change in adhesion/spreading of RAW264.7 macrophages in response to the immune stimulant lipopolysaccharide (LPS). This method enables selective, reversible, and quantitative long-term impedance measurements on single cells. The fully sealed electrofluidic assembly is compatible with long-term cell culturing, and could be modified to incorporate single cell lysis and subsequent intracellular separation and analysis.
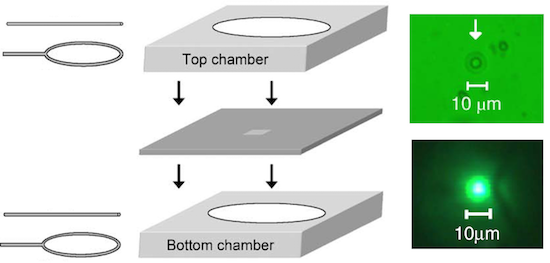
Living cells synthesize and utilize femtomole and picoliter amounts of material, and an important goal of analytical chemistry is to develop artificial interfaces to efficiently study substances on this scale. This could be achieved with a picoliter container that could be controllably loaded, transported, and unloaded, most desirably in a microfluidic environment. Phospholipid vesicles – surfactant multilayers that can form 10 μm spheres – have been studied for this purpose, but they suffer from fragility and high deformability, which have made them difficult to handle and have limited their application. We present an approach in which a gel is formed in vesicles shortly after they are created. Microfluidic mechanical testing of these vesicles shows that, in the absence of gel, vesicles are difficult to maintain in a trapped state, but the reinforced vesicles exhibit a wide window of pressures under which they can be trapped and manipulated. This improvement is likely to be an essential feature of practical applications of vesicles as microfluidic cargo containers.
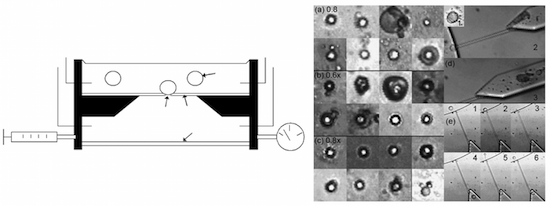
We present a new way to transport and handle picoliter volumes of analytes in a microfluidic context through electrically monitored electroporation of 10–25 μm vesicles. In this method, giant vesicles are used to isolate analytes in a microfluidic environment. Once encapsulated inside a vesicle, contents will not diffuse and become diluted when exposed to pressure-driven flow. Two vesicle compositions have been developed that are robust enough to withstand electrical and mechanical manipulation in a microfluidic context. These vesicles can be guided and trapped, with controllable transfer of material into or out of their confined environment. Through electroporation, vesicles can serve as containers that can be opened when mixing and diffusion are desired, and closed during transport and analysis. Both vesicle compositions contain lecithin, an ethoxylated phospholipid, and a polyelectrolyte. Their performance is compared using a prototype microfluidic device and a simple circuit model. It was observed that the energy density threshold required to induce breakdown was statistically equivalent between compositions, 10.2 ± 5.0 mJ/m2 for the first composition and 10.5 ± 1.8 mJ/m2 for the second. This work demonstrates the feasibility of using giant, robust vesicles with microfluidic electroporation technology to manipulate picoliter volumes on-chip.

Reversible tubing connections for glass micro-fluidic systems are realized using electro-chemical discharge machining of three-dimensional glass vias. The connections reversibly connect standard sized plastic tubing to holes in borosilicate microscope slides. Tubing connections are demonstrated on a sealed, micro-fluidic channel which is fabricated between two glass slides using SU-8. The connections are experimentally tested to withstand up to 30 psi (~206 kPa) of air pressure without leaking.
